Emissions have dominated the environmental conversation lately, which is all sorts of alright. If we don’t get a handle on what we pump into the atmosphere, our planet will become inhospitable to a lot of the people who live on it. But emissions are not the only bear we have to face down. Plastic is high on my personal 🚨 list, and the good news is, it’s easier for regular people like us to make an impact here. Packaging—that bag of chips, the blister pack around your sweet new stapler—makes up 46 percent of plastic waste. There are easy ways we can cut our plastic use, so buckle your save-the-world belts because we’re gettin’ after it.
But first, as my shoe repair guy says, you have to know what you’re fighting. When I started digging into this subject, I felt a splinter in the back of my mind: Did I actually know what plastic was? Do other people know? I started asking around:
My dad: “It’s oil.”
My friend, Brian: “I need to read more before I answer that question.”
My wife: “a synthetic compound made up of organic molecules.”
My neighbor, Anne: “often unnecessary.”
I asked more than four people, but those responses represent the spread. And as for my own answer: something something polymer triangle of rotating arrows. Thank god the scientists still take my calls, because, as with most questions involving materials science, the answer is complicated. I’m gonna break it down, and you’re gonna share this with everyone you know who uses plastic.
Let’s set some expectations: I’m not here to slag or praise plastic today, though either is easy to do. Plastic is a resource-intensive product that chokes out our oceans and ecosystems; it is no friend to dolphins, and contaminates our food supply. Annnnnd: Plastic enables everything from shelf-stable kitchen staples to space exploration to life-saving surgeries to YouTube.
Plastic is your brilliant friend who can be kind of an asshole.
It’s worth spending a little time on the word “plastic,” too. It comes from the Greek word, “πλαστικος” (“plastikos”), which means “fit for molding.” This dates back to Plato’s Timaeus, in which the eponymous character, Timaeus of Locri, delivers a hundred-ish-page monologue about the composition of the natural world and the origins of the universe. I torched half a day reading it to extract the two sentences that are relevant to this subject:
Let us proceed to distribute the elementary forms, which have now been created in idea, among the four elements. To earth, then, let us assign the cubical form; for earth is the most immoveable of the four and the most plastic of all bodies, and that which has the most stable bases must of necessity be of such a nature.
If I’ve got my Plato right (BIG IF), Timaeus is saying that, of the four elements—earth, wind, fire, and water—earth is the most resilient because it is the most moldable.
That’s some epic and accidental foreshadowing, but the more salient origin story for plastic isn’t in ancient Greece circa 360 B.C.E.. It’s in Yonkers, circa 1907, when Leo Baekeland combined phenol and formaldehyde in an ancestor of the Instant Pot to create the first substance that didn’t exist in nature: Bakelite.
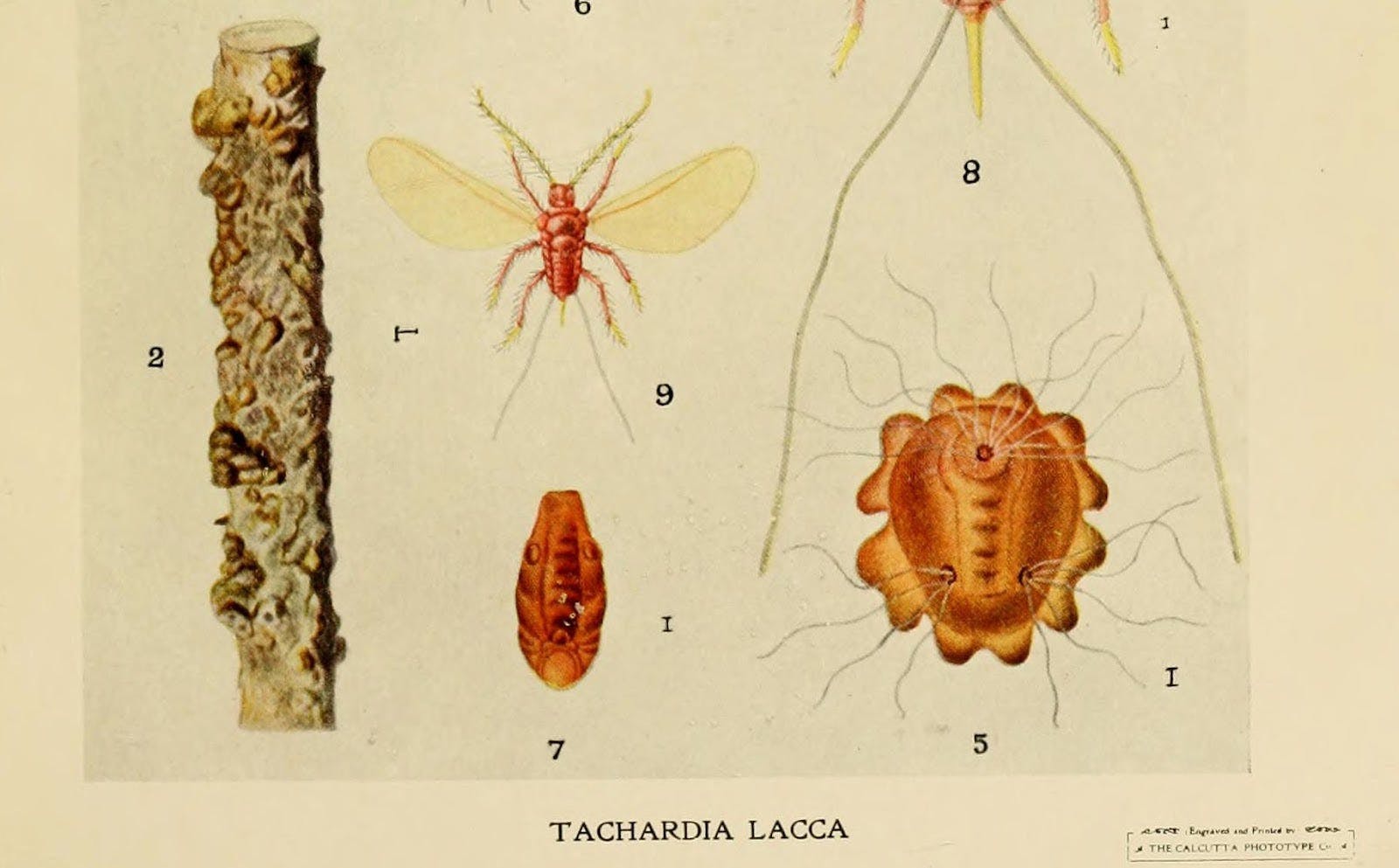
Twenty-five years earlier, Thomas Edison had opened the first central power plant in the United States. The Pearl Street Station was located in New York’s financial district, and by the time Leo got cooking, the rush was on to electrify the city. Baekeland was trying to invent a material that could replace shellac as a casing for electrical wires, because America couldn’t get enough of the stuff. Shellac, made from a resin secreted by the Southeast Asian lac bug, was (and still is) labor-intensive to manufacture and expensive to ship.
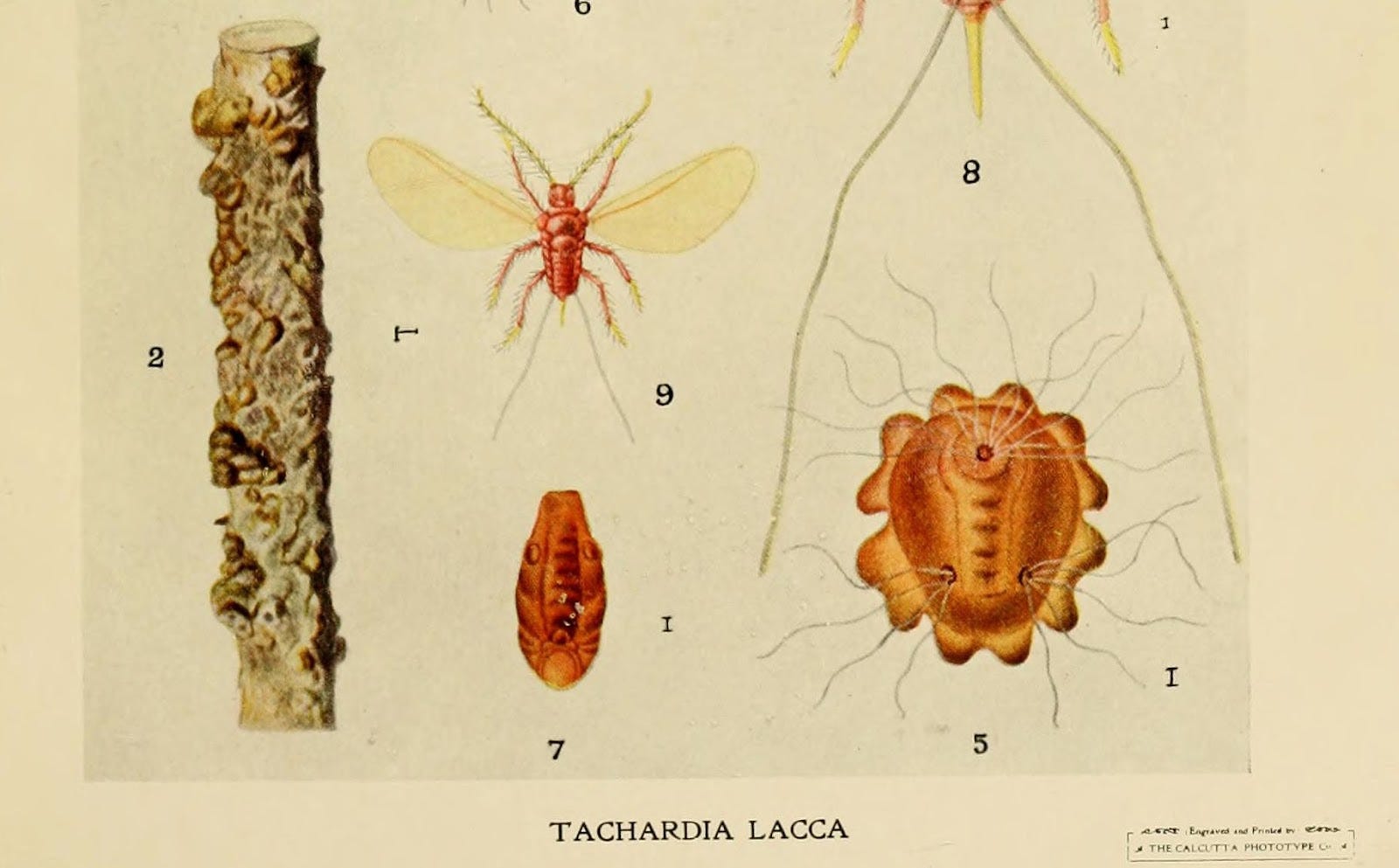
Like tortoise shell or wool or your DNA, shellac is a natural polymer. Polymers are big chains of repeating molecules; they’re tougher than other compounds due to the size of those chains. Shellac is a good electrical insulator because it’s an organic substance, and organic molecules stick together by putting electrons in a chemical straight jacket. This makes it so they don’t conduct electricity well, if at all. Shellac makes for a good wire casing because it’s moldable; and as a polymer, it’s tough. These characteristics are what Baekeland was trying to solve for, and when he pulled his tough, moldable, insulative bakelite out of the Bakelizer (real name), he called it a “plastic” and changed the world.
The difference between a naturally occurring polymer and Baekeland’s plastic is best described over breakfast. “Think about how you make a pancake,” says Scott Phillips, professor of materials science and engineering at Boise State University. “One of the main ingredients is flour, and flour is a polymer—it’s polysaccharides.” But you don’t scoop flour onto a plate and call it a flapjack. “Everything else you add to that flour gives the pancake its specific flavor and texture,” he says, noting also that a pancake is not a plastic. “Plastics are similar in that you start with your polymer, which gives you a certain amount of structure. Then you have all sorts of additives that go into it to control your desired properties.” Using various chemical components, you can tweak color, hardness, elasticity, fire resistance, and so on.
Plastic’s main chemical components have traditionally come from petroleum, but that is more about timing and convenience than scientific necessity. Baekeland made his discovery a year before Ford’s Model T debuted and began to democratize the automobile. In 1907, the U.S. only produced 166 million barrels of oil, around four percent of what we currently make, and most of it went towards producing kerosene for lighting. That process that created a ton of byproducts—like gasoline, which was often burned as waste at the refinery.
In other words, when plastics slid onto the scene, oil companies were still looking for potential applications for their sludge, and Baekeland’s discovery provided one. “The time between 1907 and WWII was the rise of the chemical industry,” says Phillips. “People were just understanding what they could mix together and what they could get out of it,” he says.
Oil provided a readily available source of hydrogen- and carbon-based organic compounds. It was “this massive supply of this super-cheap stuff that you could make chemicals from,” says Phillips. And then the military started putting in requests. “The development of plastics really took off during WWII,” he says. “The majority of polymers that we use today were invented during the war, out of necessity.”
The oil-based recipes stuck. “All of that chemistry was developed and optimized over 20 or 30 years,” says Phillips. “It’s hard to go back, once you have an optimized process and you still have that supply of oil as your starting material.” Petroleum-sourced polymers such as polyethylene, polystyrene, polypropylene, and others have enabled much of what we call modern life.
That doesn’t mean oil is the only source of polymers for plastic. “That’s something people are looking at now—a lot,” says Phillips. He cites sugar and cellulose as renewable resources that could be converted into smaller molecules, and then new kinds of polymers, and then eventually plastics. The challenge with many plant-based sources is that, unlike oil, they contain both hydrogen and oxygen, so they tend to degrade when exposed to water. (Because H₂0.)
Phillips’s lab is working on this challenge, and he and his colleagues are hitting the history books for inspiration. “Around the 1920s, Henry Ford was looking at using renewable materials to make polymers instead of using oil,” says Phillips. “His team used a mixture of phenol and formaldehyde plus soybean protein to make a plastic.” Ford even made a show car whose body panels were comprised of the soy-based stuff, and this beanmobile was 1,000 pounds lighter than an equivalent metal-bodied vehicle. Ford claimed it was better in a crash, too.
WWII derailed the project when the U.S. government turned the nation's auto factories into the makers of the war machine, and both the car and the formula for its special plastic have been lost to time.
Speaking of time, we’re out of it. If you have another minute and are blissing out in your newfound knowledge, maybe share this post with your entire internet fam. We’re @one5c on Instagram and @1fivec on Twitter. (I know, working on it.) Here are some ideas for what you can say, as well as a handy share button that is 100% reusable:
Do you actually know what plastic is? I didn’t 😬.
#TIL Ford made a car out of soybeans, but the project got mothballed when the armed forces hijacked its factories during WWII.
Both DMX and plastic were born in Yonkers.
Next week we’ll continue down the Lego brick road, but we’re gonna get more practical. I hope you’ll join me again then.
Take care of yourselves—and each other
Joe
joe@one5c.com